BiodeCCodiNNg
Decoding novel reaction chemistries in biocatalysis – Training Europe’s next visionaries for a sustainable future
We are a European research project decoding novel reaction chemistries in enzymes and thus to make organic synthesis more sustainable.
Latest Updates from BiodeCCodiNNg 📰
What is BiodeCCodiNNg about? Find out in 180 seconds!
Our publications 
Gal, Lilla; Rohan, Suresh; Żądło-Dobrowolska, Anna; Hilweg, Bianca; Müller, Judith; Tittmann, Kai; Kroutil, Wolfgang
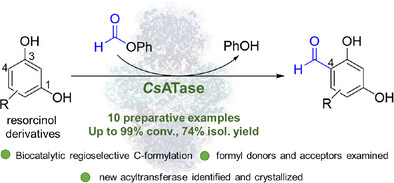
Biocatalytic Regioselective C-Formylation of Resorcinol Derivatives Journal Article
In: Angewandte Chemie International Edition, vol. 65, no. 10, pp. e19387, 2026.
@article{https://doi.org/10.1002/anie.202519387,
title = {Biocatalytic Regioselective C-Formylation of Resorcinol Derivatives},
author = {Lilla Gal and Suresh Rohan and Anna Żądło-Dobrowolska and Bianca Hilweg and Judith Müller and Kai Tittmann and Wolfgang Kroutil},
url = {https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.202519387},
doi = {https://doi.org/10.1002/anie.202519387},
year = {2026},
date = {2026-01-01},
urldate = {2026-01-01},
journal = {Angewandte Chemie International Edition},
volume = {65},
number = {10},
pages = {e19387},
abstract = {ABSTRACT Although aromatic formylation reactions are highly valuable from a synthetic perspective, a biocatalytic version has not yet been reported. Here, the cofactor-independent multimeric three-component acyltransferase from Chromobacterium sphagni (CsATase) was identified to enable the nonnatural promiscuous regioselective C-formylation of polyphenolic substrates, especially resorcinol derivatives, and thus extending the reaction scope of acyltransferases. Formylation of 4- and 5-substituted resorcinol derivatives gave access to regioselectively mono-formylated products with up to 99% conversion and up to 74% isolated yield. Formylation of phloroglucinol led to the di-formylated product with 99% conversion, outperforming chemical methods. Structural analysis of CsATase by X-ray crystallography provided insights into its active site.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Maier, Artur; Fast, Daniel; Sakalo, Dmytro; Mguni, Lindelo; Tischler, Dirk
Characterization of the N-Hydroxylating Monooxygenase TheA from Thermocrispum agreste Reveals a Broad Substrate Spectrum Journal Article
In: ChemBioChem, vol. n/a, no. n/a, pp. e202500574, 2025.
@article{https://doi.org/10.1002/cbic.202500574,
title = {Characterization of the N-Hydroxylating Monooxygenase TheA from Thermocrispum agreste Reveals a Broad Substrate Spectrum},
author = {Artur Maier and Daniel Fast and Dmytro Sakalo and Lindelo Mguni and Dirk Tischler},
url = {https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/cbic.202500574},
doi = {https://doi.org/10.1002/cbic.202500574},
year = {2025},
date = {2025-09-11},
urldate = {2025-09-11},
journal = {ChemBioChem},
volume = {n/a},
number = {n/a},
pages = {e202500574},
abstract = {The N-hydroxylating monooxygenase (NMO) TheA from Thermocrispum agreste catalyzes the N-hydroxylation step of L-ornithine, which is the first step in the thermochelin siderophore biosynthesis. Characterization of this enzyme revealed a significant thermostability up to 50 °C and activity with the non-native substrate D-ornithine with kinetic parameters (Km = 4.06 ± 0.31 mM},
keywords = {},
pubstate = {published},
tppubtype = {article}
}

Li, Yongxin; Osipyan, Angelina; Kok, Niels A. W.; Schröder, Simon; Founti, Maria; Fodran, Peter; Merkerk, Ronald; Maier, Artur; Tischler, Dirk; Schmidt, Sandy
Access to Nitrogen–nitrogen Bond-Containing Heterocycles Through Substrate Promiscuity of Piperazate Synthases Journal Article
In: ACS Catalysis, vol. 15, pp. 8846-8854, 2025.
@article{doi:10.1021/acscatal.5c01237,
title = {Access to Nitrogen–nitrogen Bond-Containing Heterocycles Through Substrate Promiscuity of Piperazate Synthases},
author = {Yongxin Li and Angelina Osipyan and Niels A. W. Kok and Simon Schröder and Maria Founti and Peter Fodran and Ronald Merkerk and Artur Maier and Dirk Tischler and Sandy Schmidt},
url = {https://doi.org/10.1021/acscatal.5c01237},
doi = {10.1021/acscatal.5c01237},
year = {2025},
date = {2025-05-11},
urldate = {2025-05-11},
journal = {ACS Catalysis},
volume = {15},
pages = {8846-8854},
abstract = {The nitrogen–nitrogen (N–N) bond motif comprises an important class of compounds for drug discovery. Synthetic methods are primarily based on the modification of N–N or N═N precursors, whereas selective methods for direct N–N coupling offer advantages in terms of atom economy and yield. In this context, enzymes such as piperazate synthases (PZSs), which naturally catalyze the N–N cyclization of l-N5-hydroxyornithine to the cyclic hydrazine l-piperazate, may allow an expansion of the current narrow range of chemical approaches for N–N coupling. In this study, we demonstrate that PZSs are able to catalyze the conversion of various N-hydroxylated diamines, which are different from the natural substrate. The N-hydroxylated diamines were obtained in situ using N-hydroxylating monooxygenases (NMOs), allowing subsequent cyclization by PZS, ultimately forming the N–N bond to yield various N–N bond-containing heterocycles. Using bioinformatic tools, we identified NMO and PZS homologues that exhibit distinct activity and stereoselectivity profiles. The screened panel yielded 17 hydroxylated diamines and more promiscuous NMOs, thereby expanding the substrate range of NMOs, resulting in the formation of previously poorly accessible N-hydroxylated products as substrates for PZS. The investigated PZSs led to a series of 5- and 6-membered cyclic hydrazines, and the most promiscuous catalysts were used to scale up and optimize the synthesis, yielding the desired N–N bond-containing heterocycles with up to 45% isolated yield. Overall, our data provides essential insights into the substrate promiscuity and activity of NMOs and PZSs, further enhancing the potential of these biocatalysts for an expanded range of N–N coupling reactions.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}

Angeli, Charitomeni; Atienza-Sanz, Sara; Schröder, Simon; Hein, Annika; Li, Yongxin; Argyrou, Alexander; Osipyan, Angelina; Terholsen, Henrik; Schmidt, Sandy
Recent Developments and Challenges in the Enzymatic Formation of Nitrogen–Nitrogen Bonds Journal Article
In: ACS Catalysis, vol. 15, no. 1, pp. 310-342, 2024.
@article{doi:10.1021/acscatal.4c05268,
title = {Recent Developments and Challenges in the Enzymatic Formation of Nitrogen–Nitrogen Bonds},
author = {Charitomeni Angeli and Sara Atienza-Sanz and Simon Schröder and Annika Hein and Yongxin Li and Alexander Argyrou and Angelina Osipyan and Henrik Terholsen and Sandy Schmidt},
url = {https://doi.org/10.1021/acscatal.4c05268},
doi = {10.1021/acscatal.4c05268},
year = {2024},
date = {2024-12-17},
urldate = {2024-12-17},
journal = {ACS Catalysis},
volume = {15},
number = {1},
pages = {310-342},
abstract = {The biological formation of nitrogen–nitrogen (N–N) bonds represents intriguing reactions that have attracted much attention in the past decade. This interest has led to an increasing number of N–N bond-containing natural products (NPs) and related enzymes that catalyze their formation (referred to in this review as NNzymes) being elucidated and studied in greater detail. While more detailed information on the biosynthesis of N–N bond-containing NPs, which has only become available in recent years, provides an unprecedented source of biosynthetic enzymes, their potential for biocatalytic applications has been minimally explored. With this review, we aim not only to provide a comprehensive overview of both characterized NNzymes and hypothetical biocatalysts with putative N–N bond forming activity, but also to highlight the potential of NNzymes from a biocatalytic perspective. We also present and compare conventional synthetic approaches to linear and cyclic hydrazines, hydrazides, diazo- and nitroso-groups, triazenes, and triazoles to allow comparison with enzymatic routes via NNzymes to these N–N bond-containing functional groups. Moreover, the biosynthetic pathways as well as the diversity and reaction mechanisms of NNzymes are presented according to the direct functional groups currently accessible to these enzymes.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}

Maier, Artur; Mguni, Lindelo M.; Ngo, Anna C. R.; Tischler, Dirk
Formate Dehydrogenase: Recent Developments for NADH and NADPH Recycling in Biocatalysis Journal Article
In: ChemCatChem, vol. 16, no. 21, pp. e202401021, 2024.
@article{https://doi.org/10.1002/cctc.202401021,
title = {Formate Dehydrogenase: Recent Developments for NADH and NADPH Recycling in Biocatalysis},
author = {Artur Maier and Lindelo M. Mguni and Anna C. R. Ngo and Dirk Tischler},
url = {https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/cctc.202401021},
doi = {https://doi.org/10.1002/cctc.202401021},
year = {2024},
date = {2024-07-24},
urldate = {2024-07-24},
journal = {ChemCatChem},
volume = {16},
number = {21},
pages = {e202401021},
abstract = {Abstract Formate dehydrogenases (FDHs) catalyze the oxidation of formate to CO2 while reducing NAD(P)+ to NAD(P)H and are classified into two main classes: metal-dependent (Mo- or W-containing) and metal-independent FDHs. The latter are oxygen-tolerant and relevant as a cofactor regeneration system for various bioprocesses and gained more and more attention due to their ability to catalyze the reverse CO2 reduction. This review gives an overview of metal-independent FDHs, the recent advances made in this field, and their relevance for future applications in biocatalysis. This includes the exploitation of novel FDHs which have altered co-substrate specificity as well as enzyme engineering approaches to improve process stability and general performance.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}

Training Europe's next visionaries for a sustainable future to create tailor-made biocatalysts
Our BiodeCCodiNNg project is a Doctoral Network (DN) that builds around a comprehensive doctoral training programme to educate Europe’s next visionaries with out-of-the-box thinking and an entrepreneurial mindset to expand the repertoire of enzymes for industrial biotechnology.
From enzyme discovery to industrial implementation – ceating novel biocatalysts for industrial and pharmaceutical biotechnology
We combine basic research and applied engineering to deliver new synthetic routes to chemically relevant products in more efficient and cleaner ways than the present ones.
Teaming up with different partners
Our network is based on a multidisciplinary team bringing together ten academic partners from renowned European universities and 10 industrial partners from all over Europe.





